Accelerating time to clinic by de-risking clone selection early in the CHO cell line development process
In addition to Cergentis' TLA-based services for Chinese Hamster Ovary (CHO) clone selection, we now offer
a new in-house solution: CHOice®.
This allows our customers to access the same high-quality results while profiting from even shorter turnaround times, cost-efficiency, scalability and confidentiality.
CHO clone selection
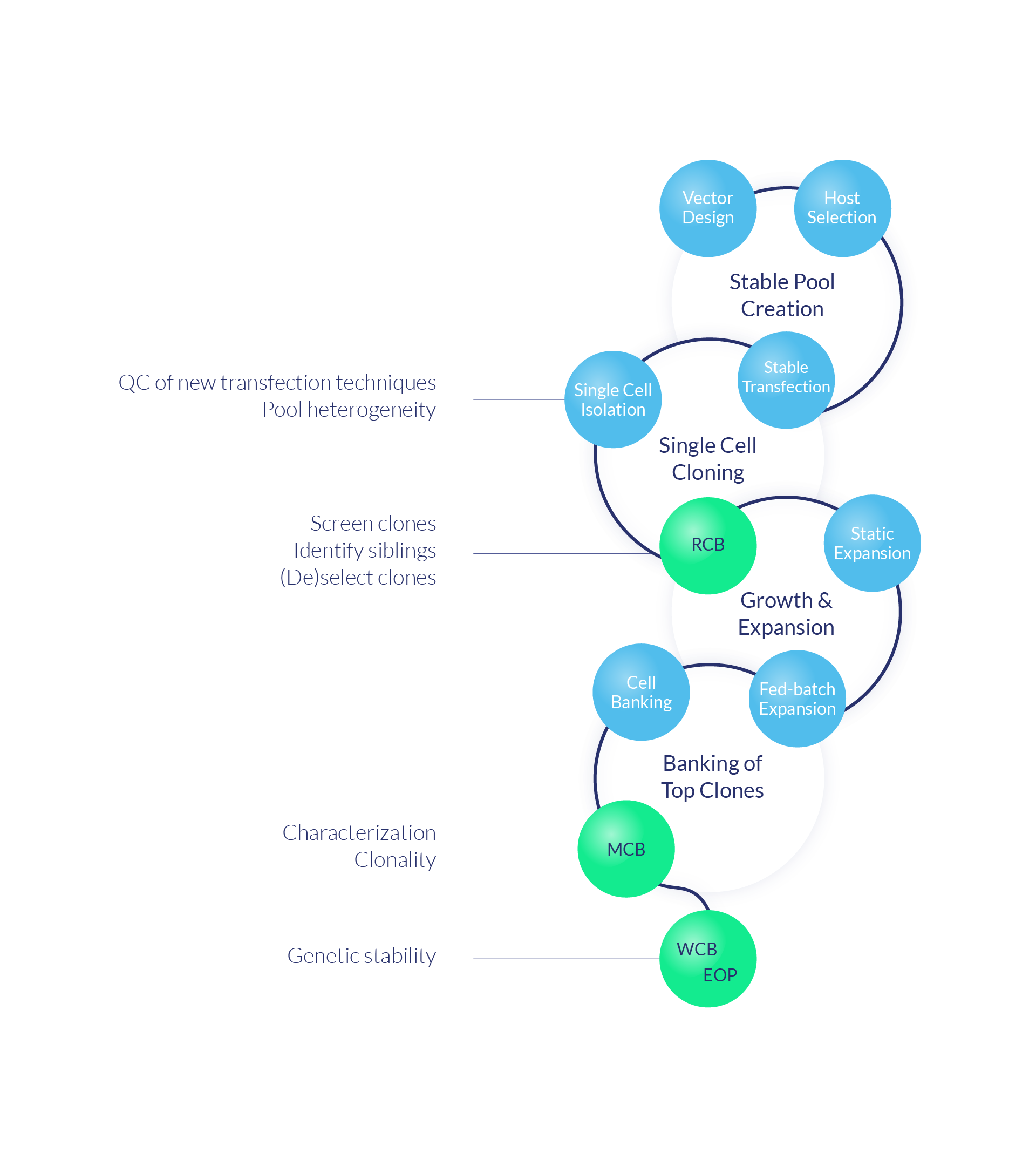
Selecting the clone with the preferred genetic make-up is a crucial step in the cell line development process toward producing stable and high-quality biotherapeutic proteins. Cergentis' TLA-based services have a proven track record in the genetic QC and characterization of cell lines and in clone selection. Conventional technologies are limited when it comes to sensitivity, cost and turnaround time. Our TLA-based analysis yields all essential genetic characteristics in a single test: an ideal platform to monitor important quality parameters for clone selection.
Identifying siblings
Deselecting clones containing undesired mutations in the coding sequence of GOI
De-risking clones early in the cell line development proces
Accelerating timelines
Facilitating early phase testing
Minimizing time to clinic
Your CHOice®
The in-house solution for CHO clone selection combines the widely-published and recognized TLA analysis with our newly developed software platform.
Integrated solution from sample processing up to analysis
More cost effective than outsourcing and at larger throughput
Reduced turnaround time (7 - 14 days from sample to results)
Master your own data and materials
User friendly and independent of Cergentis' capacity
Highly scalable
Automated, unbiased data analysis
Highest quality standard

Fully integrated solution, starting from €9,900
The fully integrated in-house solution CHOice® comprises of the following:
Reagents kit for 12 samples
User manual
Access to Cergentis' software analysis platform
Customer & tech support
Expert consultation
Orderable controls
* First time users will undergo a qualification trajectory including both wetlab and data analysis training.
Our TLA-based services for cell line development & clone selection
What our customers say
"TLA analysis has proven to be very useful to quality control genetically engineered cell lines.”
Dr. John Wiseman
Associate Director - AstraZeneca
"Working with the team at Cergentis has allowed us to add an extra dimension to the analysis of our stable cell lines. Their understanding of our needs combined with the reliability and reproducibility of TLA analysis has resulted in a number of successful projects."
Dr. Teresa Domagala
Director Manufacturing Dev. - Teva
"TLA outperforms traditional methods used in CHO cell line characterisation and provides higher resolution and sensitivity.”
Dr. Stefanie Bartels
Project Leader CLD - Byondis


